Table of Contents
Retatrutide: The Triple Agonist Revolutionizing Weight Loss in 2026 – Full Deep Dive Update
As of March 2026 in Winnipeg’s still-chilly spring, the obesity treatment landscape is evolving faster than ever. Retatrutide (Eli Lilly’s investigational LY3437943) stands out as the most potent contender yet — a first-in-class triple hormone receptor agonist targeting GLP-1, GIP, and glucagon pathways. This multi-action mechanism delivers superior appetite suppression, improved insulin sensitivity, boosted energy expenditure, and enhanced fat burning compared to dual agonists like tirzepatide or single GLP-1s like semaglutide.
The buzz exploded in December 2025 with the first Phase 3 topline results from the TRIUMPH-4 trial. Since then, anticipation has built for the remaining seven Phase 3 readouts expected throughout 2026. Here’s the most comprehensive, up-to-date breakdown: mechanisms, trial data, comparisons, side effects, timeline, and real-world implications for sustainable health in places like Manitoba.
Understanding Retatrutide: How the Triple Mechanism Works
Retatrutide mimics three gut hormones simultaneously:
- GLP-1 — Reduces appetite, slows gastric emptying, promotes satiety.
- GIP — Enhances insulin release, supports fat metabolism.
- Glucagon — Increases energy expenditure (calorie burn at rest), mobilizes fat stores, and prevents metabolic slowdown during weight loss.
This “triple hit” addresses common plateaus seen in earlier drugs by ramping up metabolism while curbing hunger. Administered as a once-weekly subcutaneous injection (similar to Zepbound or Wegovy pens), it’s designed for long-term use alongside diet and exercise.
(Example of a similar once-weekly injection pen device used for incretin-based therapies like tirzepatide.)
(Another view of a typical GLP-1/GIP injection pen — retatrutide’s delivery will likely resemble this.)
Breaking Down the Landmark TRIUMPH-4 Phase 3 Results (December 2025)
In TRIUMPH-4 (adults with obesity/overweight + knee osteoarthritis, no diabetes):
- Duration: 68 weeks.
- Doses tested: 9 mg and 12 mg vs. placebo (all with lifestyle counseling).
- Key outcomes (completers analysis):
- 12 mg: 28.7% average body weight loss (~71.2 lbs from ~248 lb baseline).
- 9 mg: 26.4% loss (~64 lbs).
- Placebo: 2.1% loss.
- Pain reduction: Up to 75.8% drop in WOMAC pain scores for knee osteoarthritis — a huge bonus for joint health.
- Other wins: Improved physical function, cardiovascular risk markers.
- Responder rates: High percentages achieved ≥20–35% loss.
Even intent-to-treat (including dropouts) showed strong placebo-adjusted results (~23–26%). No plateau was observed; loss continued progressively.
Phase 2 data laid the foundation: ~17.5% at 24 weeks, ~24.2% at 48 weeks on top doses.
Retatrutide vs. Current Leaders: 2026 Comparison Table
| Medication | Targets | Avg. Max Weight Loss (Phase 3) | Key Trial Duration | Status (March 2026) | Standout Benefits / Notes |
|---|---|---|---|---|---|
| Semaglutide (Wegovy/Ozempic) | GLP-1 only | 15–20% | 68–72 weeks | Approved | Proven safety, widely available; solid but lower ceiling. |
| Tirzepatide (Zepbound/Mounjaro) | Dual GLP-1 + GIP | 20–22.5% | 72 weeks | Approved | Current benchmark; beats semaglutide head-to-head. |
| Retatrutide | Triple GLP-1 + GIP + Glucagon | Up to 28.7% (TRIUMPH-4) | 68 weeks | Phase 3 ongoing | Highest reported; glucagon adds metabolic boost; potential for 25–30%+ in broader populations. |
For a 250-lb starter: Retatrutide could mean 70+ lbs lost vs. 50–56 lbs on tirzepatide.
Side Effects Profile: Balanced View from Latest Data
Mostly gastrointestinal (GI), dose-dependent, and often improving over time:
- Common: Nausea, vomiting, diarrhea, constipation.
- Other: Temporary heart rate increase (peaks early, then declines), fatigue, headache.
- New signal in TRIUMPH-4: Dysesthesia (abnormal/painful touch sensations) in ~9–21% on active doses (vs. <1% placebo) — not a major discontinuation driver but needs monitoring.
- Dropout rates: Higher at top doses (some cited “too much” loss or GI issues).
- No severe hypoglycemia or new major red flags; profile similar to tirzepatide but potentially more intense GI at peak doses.
Long-term safety awaits full 2026 data.
Expected Timeline and What’s Next in 2026
- Not yet approved; still investigational.
- Seven more TRIUMPH trials readout in 2026 (obesity core, diabetes, sleep apnea, maintenance dosing including 4 mg, cardiometabolic outcomes, etc.).
- If consistent/safe: Potential FDA submission late 2026 → approval possibly 2027.
- Meanwhile: Focus on approved options + lifestyle (strength training, walking in Winnipeg’s indoor options like heated classes).
Visualizing the Impact: Real Transformation Examples
While retatrutide-specific before/after photos from trials aren’t publicly released yet (due to ongoing studies), similar incretin therapies show dramatic changes. Here are illustrative examples of significant weight loss transformations:
(Real-world example of substantial body composition change after peptide-assisted weight loss.)
(Dramatic before-and-after showing obesity to athletic build — highlights potential with high-efficacy agents.)
(Another patient transformation: Noticeable reduction in abdominal fat and improved posture/mobility.)
(Inspiring shift from higher body weight to active, confident lifestyle.)
(Classic side-by-side mirror progress shot common in weight loss journeys.)
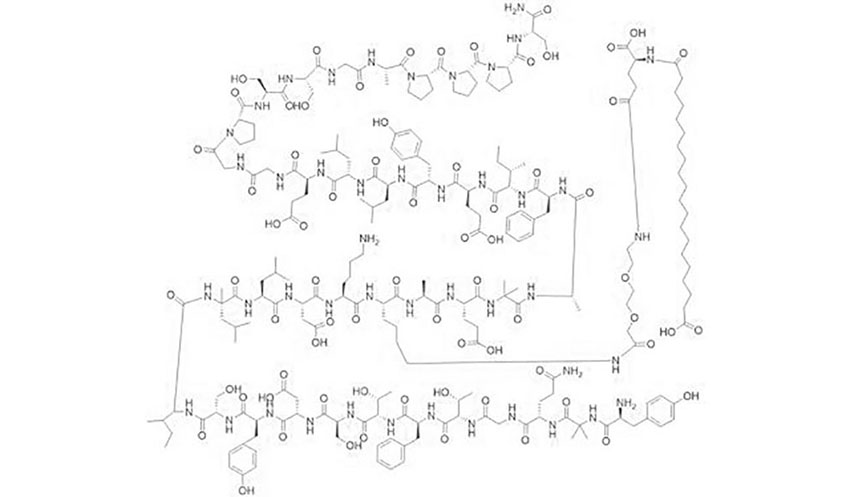
Retatrutide’s Molecular Structure (For the Science Lovers)
(One representation of retatrutide’s complex peptide structure.)
(Detailed chemical diagram showing the triple-agonist design.)
Why Retatrutide Could Change Everything for Long-Term Health
In 2026, the goal isn’t just pounds lost — it’s reversing obesity complications like joint pain, metabolic issues, and cardiovascular risk. Retatrutide’s triple action + osteoarthritis pain relief positions it uniquely. Pair it with Winnipeg-friendly habits: indoor strength sessions, walking challenges, protein-focused meals, and recovery focus.
Excited for the rest of 2026’s data drops? This could redefine what’s “possible” in non-surgical weight management.
What excites you most about retatrutide — the weight loss potential, pain relief, or metabolic boost? Share in the comments!
(Not medical advice — consult your doctor. Info based on public trial announcements as of March 2026.)




:max_bytes(150000):strip_icc():focal(749x0:751x2)/linda-migliaccio-1-2086c9c13aff44d2aed0a852353cc4fd.jpg)